Coming Soon: GMP-Grade* Recombinant Monoclonal Antibody Conjugates for Cell Therapy Manufacturing & QC
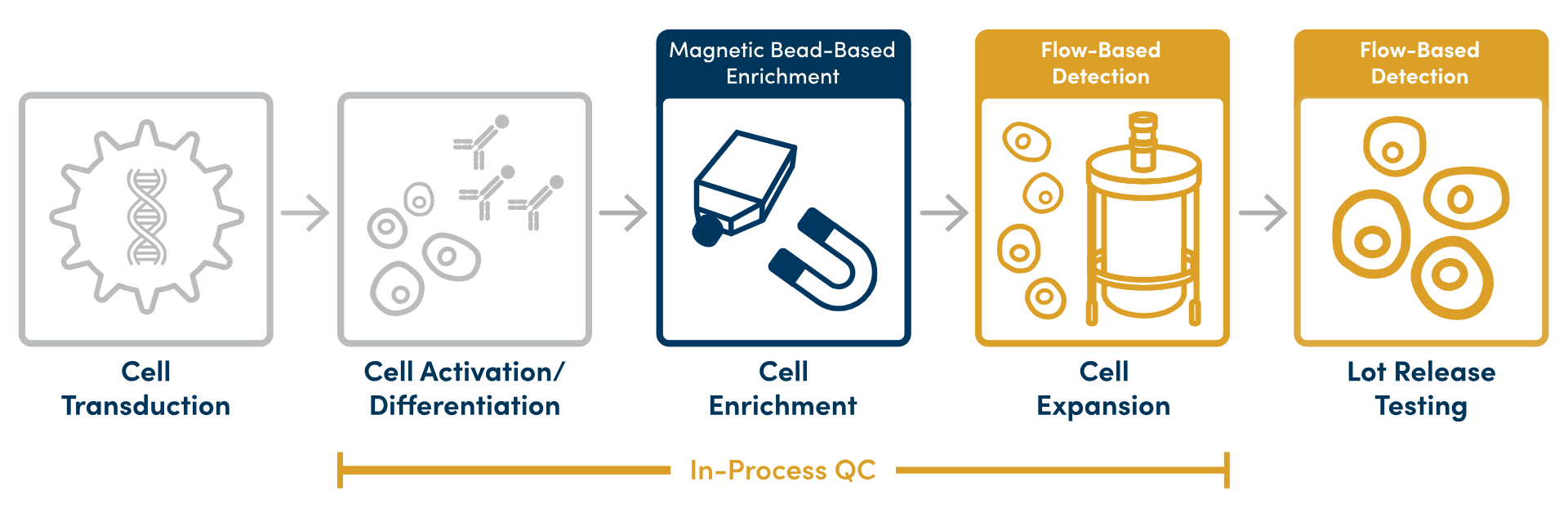
Incorporate robust, flexible CAR-T Cell Enrichment and Detection into Your Manufacturing and QC Workflows
CST revolutionized preclinical CAR cell characterization with universal recombinant monoclonal antibodies specific to the ubiquitous Whitlow/218 and G4S linker sequences of scFv-based CARs. This innovation enabled scientists to use a single reagent for a variety of assays without the time or cost of developing anti-idiotype antibodies for every CAR construct.
When available, these trusted antibody conjugates, manufactured to be GMP-grade*, are intended to simplify enrichment of G4S linker CAR+ cells and increase productivity for in-process QC and lot release assays. When preclinical development teams use the same reagents, handoffs can be that much easier as programs advance to later stages.
| CAR+ Cell Enrichment in Manufacturing | |
Coming soon: GMP-Grade* G4S Linker (E7O2V) Rabbit Monoclonal Antibody (Biotin Conjugate)
| |
| CAR+ Cell Detection for Analytical QC | |
Coming soon: GMP-Grade* G4S Linker (E7O2V) Rabbit Monoclonal Antibody (PE Conjugate)
| |
| Coming soon: Seamless Transfer from Preclinical to Clinical* | |
| |
| Reagent Security and Supply Continuity | |
| |
* GMP-grade antibody reagents are under development and not currently available. The currently-available pre-GMP antibody reagents are not yet validated for GMP, are for Research Use Only (RUO) and are not for use in clinical manufacturing stages. Full GMP-grade certification and commercial quantities are expected to be available mid-2026.